Help Protect Yourself: Get Screened and Vaccinated
HPV is one of the most common sexually transmitted infections (STIs) in Canada and is responsible for nearly 100% of cervical cancer cases. Depending on your province and if you’re sexually active, regular cervical cancer screening through Pap or HPV testing can start at age 21 or 25.
In some provinces, Pap tests are being replaced by HPV testing as the primary method for cervical cancer screening.
HPV testing can detect the high risk strains of HPV that may cause cervical cancer and related cell changes. It can detect HPV even before cell changes happen so that finding and treating problems can happen earlier, often before cancer develops.

HPV Test: What It Is and Why It’s Important
Self-screening for the HPV virus is now available in provinces like British Columbia and Prince-Edward-Island, giving you the option to test on your own or with a healthcare provider. These tests are accessible, accurate and affordable.
Understand the difference
HPV Test vs Pap Test
HPV Test (Cervical Screening Test)
An HPV test can be performed by a healthcare professional (family doctor, OBGYN or nurse) in a doctor’s office or clinic.
Self-screening via mail is in certain provinces and can be performed in the comfort of your home. Some doctors’ offices and clinics may also offer self-testing on site.
Pap Test
Pap tests are performed by a family doctor, OBGYN or nurse at a doctor’s office or clinic.
HPV Test (Cervical Screening Test)
The HPV test is a vaginal test that looks for the presence of high-risk HPV. It is very accurate and is becoming an important first step in cervical screening.
Pap Test
A Pap test can help detect abnormal cells in the cervix that are cancerous or may have the potential to become cancerous.
A Pap test may be provided as a standalone or after a positive HPV test.
HPV Test (Cervical Screening Test)
The HPV test identifies if a person is HPV positive or negative. Like a Pap test, collection is still performed with a swab vaginally but the sample testing is carried out differently.
Pap Test
Cells collected from the cervix and examined under a microscope.
HPV Test (Cervical Screening Test)
- Detects HPV before cell changes occur
- Can identify pre-cancerous conditions earlier
Pap Test
- Detects existing abnormal cells
- Helps identify pre-cancerous or cancerous changes
What do my results mean?
If no HPV is found:

Following your screening, no further testing is needed for 3 to 5 years, as the likelihood of detecting abnormal cervical cells is minimal.
If HPV is found:

A healthcare professional will guide you through the next steps. This does not mean you have or will develop cancer. Additional testing and check-ups will likely be suggested by your healthcare professional so they can keep an eye on your cervical health.
Why HPV vaccination matters
Getting the HPV vaccine helps protect you from certain types of HPV that cause most cervical cancers. However, since the vaccine doesn’t protect against all types of HPV, regular screening and practising safer sex are still important.
An estimated 75% of sexually active Canadians will have at least one HPV infection in their lifetime.

Have an open conversation with your partner
HPV can affect anyone who’s sexually active—not just women. Because there’s no routine HPV test for men yet, it’s important to talk openly with your partner about protection.
Did you know?
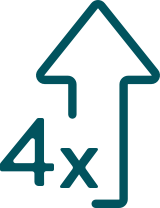
In 2012, men were more than 4 times more likely than women to develop certain HPV-related cancers in the mouth and throat (oropharyngeal cancers or OPC).

When you’re already seeing your healthcare provider for something like a UTI, yeast infection, or birth control, it’s a good opportunity to discuss HPV prevention, including vaccination options for you and your partner.
Canada and HPV testing access
Provinces and territories across Canada are expanding and updating access to HPV testing. You can find more information about cervical cancer screening through the links below.
Safety Information
GARDASIL®9 is a vaccine indicated for individuals 9 through 45 years of age for the prevention of infection caused by the human papillomavirus (HPV) types 6, 11, 16, 18, 31, 33, 45, 52 and 58 and the following diseases associated with the HPV types included in the vaccine: certain head and neck cancers, such as throat and back of mouth cancers, caused by HPV types 16, 18, 31, 33, 45, 52, and 58. GARDASIL®9 has been approved with conditions. This means it has passed Health Canada’s review and can be bought and sold in Canada, but the manufacturer has agreed to complete more studies to make sure the vaccine works the way it should. For more information, talk to your healthcare professional.
GARDASIL®9 is also indicated for individuals 9 through 45 years of age for the prevention of infection caused by the human papillomavirus (HPV) types 6, 11, 16, 18, 31, 33, 45, 52 and 58 and the following diseases associated with the HPV types included in the vaccine: cervical, vulvar, vaginal and anal cancer caused by HPV types 16, 18, 31, 33, 45, 52, and 58; genital warts (condyloma acuminata) caused by HPV types 6 and 11; abnormal and precancerous cervical lesions (changes in cells of the cervix that have a risk of turning into cancer) as found in a Pap test caused by HPV types 6, 11, 16, 18, 31, 33, 45, 52 and 58; abnormal and precancerous vaginal, vulvar (outside of the female genital area) and anal lesions caused by HPV types 6, 11, 16, 18, 31, 33, 45, 52 and 58.
GARDASIL®9 does not protect the person getting the vaccine from a disease that is caused by other types of HPV, other viruses, or bacteria, and from HPV types that he/she may already have; but most people do not have all types contained in the vaccine. GARDASIL®9 does not treat HPV infection. GARDASIL®9 does not remove the need for screening for cervical, vulvar, vaginal, anal, and certain head and neck cancers, such as throat and back of mouth cancers; women should still get routine cervical cancer screening. You should not get GARDASIL®9 during pregnancy. If you plan to get pregnant or get pregnant, you should wait until you are no longer pregnant to complete your vaccine series. In more than 1,000 pregnancies, women who were pregnant or became pregnant after getting GARDASIL®9 did not have a higher chance for miscarriages or babies with birth defects. Pregnant women exposed to GARDASIL®9 are encouraged to report their exposure or suspected adverse reactions by contacting Merck Canada Inc., at 1-800-567-2594. It is not known whether GARDASIL®9 is excreted in human milk. If you are breast-feeding, talk to your doctor or health care professional to see if you should be vaccinated with GARDASIL®9. Tell the doctor or health care professional if you or your child (the person getting GARDASIL®9), is pregnant or is planning to get pregnant. GARDASIL®9 may cause some side effects and allergic reactions. The most common side effects seen are pain, swelling, redness, itching, bruising, bleeding, a lump where the shot is given, headache, fever, nausea, dizziness, tiredness, diarrhea, abdominal pain, and sore throat. Fainting can happen after getting an HPV vaccine. Signs of an allergic reaction may include difficulty breathing, wheezing (bronchospasm), hives and rash. This is not a complete list of side effects; for any unexpected effects while taking GARDASIL®9, contact your doctor or pharmacist. Contact your doctor or healthcare professional to determine if GARDASIL®9 is an appropriate option for you.
Information about GARDASIL®9 is provided in the Product Monograph and Consumer Information, available here.
HPV=human papillomavirus.